FDA Expands Approval for Emergent BioSolutions' Smallpox Vaccine Amid Mpox Crisis
The U.S. FDA has expanded approval for Emergent BioSolutions' smallpox vaccine, ACAM2000, for high-risk mpox individuals, boosting the company's shares by 20%. Despite its restrictions for people with weakened immune systems, ACAM2000 is the second approved mpox vaccine in the U.S. As mpox cases surge in Africa, Emergent will donate 50,000 doses to affected regions.
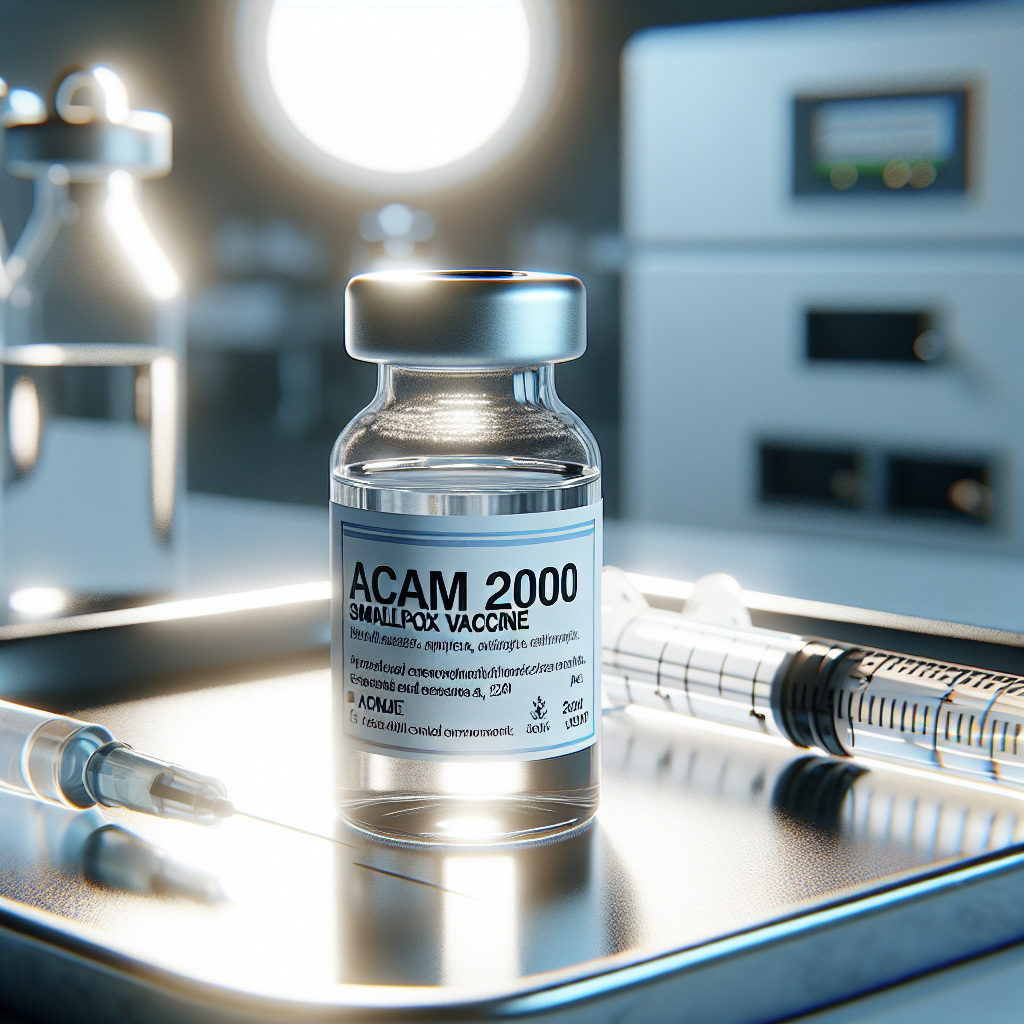
The U.S. Food and Drug Administration granted expanded approval to Emergent BioSolutions' smallpox vaccine for people at high risk of mpox infection, boosting the drugmaker's shares by 20% pre-market on Friday.
The FDA's clearance, announced late Thursday, authorizes the vaccine, ACAM2000, as the second mpox vaccine in the U.S. after Bavarian Nordic's Jynneos. However, ACAM2000 is unsuitable for individuals with weakened immune systems, including those with HIV. Earlier, during a 2022 U.S. mpox outbreak, its usage was limited despite being part of the national stockpile.
Both Jynneos and ACAM2000 contain the vaccinia virus, related to but less harmful than the smallpox and mpox viruses. Notably, ACAM2000 utilizes a live, infectious form of the vaccinia virus. This approval comes amid the rapid spread of a new mpox strain, clade Ib, in Africa, prompting the World Health Organization to declare mpox a global public health emergency this month.
"Mpox has evolved into an uncontrolled epidemic in Africa, necessitating all effective tools to eliminate it as a threat," said Dr. Amesh Adalja of the Johns Hopkins Center for Health Security. Last week, Emergent BioSolutions pledged to donate 50,000 doses of its smallpox vaccine to the Democratic Republic of the Congo and other affected countries like Burundi, Kenya, Rwanda, and Uganda.
Gaithersburg, Maryland-based Emergent BioSolutions experienced a share slump last year due to weakness in its contract manufacturing business. However, shares have tripled this year following job cuts and the appointment of former Bausch CEO Joseph Papa to spearhead the company's turnaround.
(With inputs from agencies.)
- READ MORE ON:
- FDA
- Emergent BioSolutions
- smallpox
- mpox
- vaccine
- ACAM2000
- public health
- WHO
- Africa
- global emergency
ALSO READ
Health Updates: Polio in Gaza, COVID and Mpox Vaccines, Opioid Settlement, and More
Gaza's Children Receive Polio Vaccines Amidst Ongoing Conflict
UNICEF Issues Emergency Tender for Mpox Vaccines
Health News Roundup: Nigeria's Mpox Vaccination Rollout, Novavax's COVID Vaccine, and More
UNICEF Launches Emergency Tender for Monkeypox Vaccines










